
How to fill out Solomon's soil analysis worksheets
 Before I dive any deeper into
soil science, I thought I'd walk you through filling out Solomon's soil
analysis worksheets (which you can download
for free here).
You'll notice there are actually six pages of worksheets in that file,
which consist of two pages each for acidic soils (pH less than 7),
"excess cations" soil (pH 7 to 7.6), and calcareous soil (pH greater
than 7.6). I actually find it much easier to make a
spreadsheet page for each soil sample since the program can do the math
for me, but I'll fill out a worksheet below to help you get an idea of
the process.
Before I dive any deeper into
soil science, I thought I'd walk you through filling out Solomon's soil
analysis worksheets (which you can download
for free here).
You'll notice there are actually six pages of worksheets in that file,
which consist of two pages each for acidic soils (pH less than 7),
"excess cations" soil (pH 7 to 7.6), and calcareous soil (pH greater
than 7.6). I actually find it much easier to make a
spreadsheet page for each soil sample since the program can do the math
for me, but I'll fill out a worksheet below to help you get an idea of
the process.
Since I sprang for a
test from Logan Labs, as
Solomon recommended,
it's pretty simple to fill out the column of actual amounts. The
only tricky parts are:
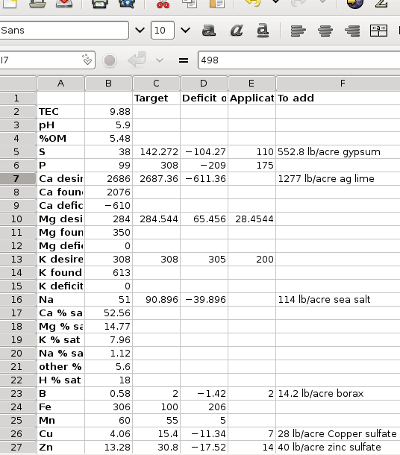
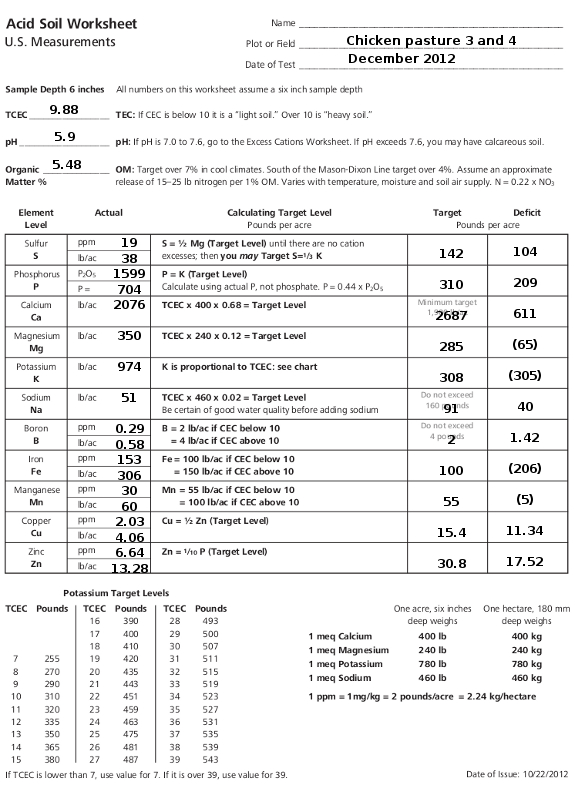
- You need to convert from ppm (parts per million) to lb/acre (pounds per acre) for certain readings. Solomon explains that you simply multiply ppm by 2 to get lb/acre, which I'm a little dubious about. His reasoning is that we sampled our soil to a six inch depth, and soil scientists estimate that amount of earth weighs about two million pounds per acre. When I start cancelling units in the conversion, though, I feel like there should be something factored in to take the atomic weight of each mineral into account, but I stuck to Solomon's math. (Roland, help?)
- Logan labs reports phosphorus pentoxide instead of elemental phosphorus, so you need to multiply their result by 0.44 to get lb/acre for phosphorus.
The target column is a
little more complex, but is mostly basic multiplication. The one
portion that might cause a hiccup is potassium (K) --- you get that
amount from the chart at the bottom-left of the worksheet based on the
TCEC of your sample. Similarly, boron, iron, and manganese
targets are based on TCEC, as is explained in the "calculating target
level" column.
 Finally, you subtract the
actual amount of each element (in lb/acre) from the target amount to
figure out how much excess or deficient you are. Since Solomon
labelled the last column "deficit", I put excesses in parentheses.
Finally, you subtract the
actual amount of each element (in lb/acre) from the target amount to
figure out how much excess or deficient you are. Since Solomon
labelled the last column "deficit", I put excesses in parentheses.
The sample I used is a
pasture that has been grazed with chickens for a couple of years with
no other amendments, so I figure it's probably similar to the soil you
might find in a new garden spot. You'll notice the soil is acidic
and a bit low on organic matter, without as much capacity for cations
as you'd like, and it has too much of a few nutrients but too little of
some others. Tomorrow, I'll move on to the back side of this
worksheet to show you how to deal with those excesses and deficits.
This
post is part of our The Intelligent
Gardener lunchtime series.
Read all of the entries:
|
Want more in-depth information? Browse through our books.
Or explore more posts by date or by subject.
About us: Anna Hess and Mark Hamilton spent over a decade living self-sufficiently in the mountains of Virginia before moving north to start over from scratch in the foothills of Ohio. They've experimented with permaculture, no-till gardening, trailersteading, home-based microbusinesses and much more, writing about their adventures in both blogs and books.
Want to be notified when new comments are posted on this page? Click on the RSS button after you add a comment to subscribe to the comment feed, or simply check the box beside "email replies to me" while writing your comment.

When we are talking about ppm, we first need to know what exactly it means in this context, since it is a fraction of two measurements that have the same unit and is therefore dimensionless. E.g. ppm can be an elongation (in/in), solution by weight (lb/lb) or fraction by particles (e.g. mole/mole).
Assuming that the report is in lb/lb and not in mole/mole, we need to figure out the dry bulk density of soil. We use the dry density because we are interested in the mass ratios of the solids in the soil. The particle density of minerals varies a bit, but not much. The dry bulk density of soil can vary a lot depending on the exact mineral composition, the amount of organics and the degree of compaction. It is generally reckoned to be between 1.0--1.6 kg/dm³.
Six inches is 1.524 dm. One acre is 404685.64 dm². So this soil would have a volume of 616741 dm³. With the given dry bulk density range, you would have a dry soil weight of 616741--986786 kg, or 1359681 to 2175490 lb. So under the abovementioned assumptions, 1 ppm would correspond to between 1.36 and 2.18 lb/acre.
Hi Anna,
One consultant suggests 3 ppm of Mo so the soil microbes work well.
And humans do better with some V, Se, Ag, Cr.
Once you have added greensand and seaweed to somewhere and it has settled in a more detailed soil report would probably be well worth it :).
I gather you have no problem with having enough water :).
:).
John
I'm not finding any Excel worksheets that correspond to the text descriptions on this website or in the addendum IntelligentGardener-worksheets.pdf to which you link above. Are these Excel worksheets really available? I've been attempting to determine if I have enough information to create some of my own, but it would save me a lot of time if such were downloadable. Thanks, db
Anna, thanks for your reply. I'm looking at the figures on this page. I'm not able to arrive at the deficit for K. I see you took 308 from the table (corresponding to TCEC = 9.88) but what is the actual figure from which you subtract 308? Lb/ac = 974, so I calculate a deficit of -666. But your spreadsheet shows a deficit of -305. I must be missing something. I read all of the article without understanding. I see that cell B14 in the spreadsheet shows 613 as a found amount, but the soil analysis shows K = 974. Is that the difference? (Substituting 613 for 974 provides the same result as entered in the worksheet.) In case you have time to reply - Thanks! db
The revised worksheets have been revised and are at http://soilanalyst.org/wp-content/uploads/2012/12/WorksheetRevision-03.pdf This note from Erica Reinheimer: We have recently cut the P rate in half (to allow for future high P compost additions) - this change does not yet appear in the worksheets. Erica also send a revised table of TCEC target levels. (I don't see a way to attach it here. She's erica@growabundant.com) in case you're interested. Thanks for the replies! Regards, db
Very nice work. I note you said you had worked up the excel sheets. Please share them. I will convert them to metric and go through the calculations, I am a soil scientist so may pick up on any error you may have made, but will likley make my own. Quick check TCEC stands for Total Cation Exchange Capacity yes ? Also I am presuming you are considering temperate rather than tropical soils. I would like to do similar for tropical soils for here in Zambia. regards Ben
I also have created spreadsheets to automate the calculation of soil amendments. I have used them with success in three garden areas. I use the results to provide minerals to my gardens according to the Rev 03 worksheets from Erica Reinheimer and Steve Solomon. I do not guarantee or warrant my workbook to be free of errors or necessarily suitable for use by anyone else. I recommend that you fully understand the principles and practices detailed in the Rev 03 worksheets. My calculated results either match or or close to those generated by OrganiCalc. I believe I am more careful to define the specific mineral contents of the amendment materials I use. I offer this Excel workbook for your potential learning or use at your own risk. A zip archive is available for download from http://hillhousehome.net/Gardening/AcidSoil.zip.
Greetings,
We are continually improving the methods Steve Solomon and I wrote in 2012 in "The Intelligent Gardener - Growing Nutrient Dense Food" We document the changes to our OrganiCalc worksheet, and every year or 2 release the changes as a hardcopy worksheet. There are many free apps on our site, and the latest release of OrganiCalc is still available for $9.50/year. I like to make comments on the results as time permits.